All published articles of this journal are available on ScienceDirect.
Challenges in the Diagnosis of Invasive Cribriform Cancer of the Breast: A Case Report and Literature Review
Abstract
Background
Invasive cribriform cancer of the breast (ICC) is a rare type of breast cancer characterized by its unique cribriform cell pattern. It is frequently seen in menopausal women, presenting with bloody nipple discharge or breast mass as an incidental finding. It is classified as a good prognosis tumor; however, it imposes diagnosis challenges due to its shared similarities with other breast cancers and coexistence with various histological subtypes.
Case Presentation
A 62-year-old lady presented with a 2-month history of breast discharge with a palpable mass. An imaging study revealed a BI-RADS 4 category (according to imaging reporting and data system), implying a definitive probability of malignant pathology. A core needle cytology suggested atypical hyperplasia or invasive malignancy; an excisional biopsy and immunohistochemistry study confirmed ICC diagnosis.
Discussion
The integration of histopathological examination with immunohistochemistry unveiled ICC diagnosis and excluded other differential diagnoses. Several challenges can be associated with ICC diagnosis, as it may present with subtle or atypical imaging criteria or even tend to regress, which is why radiologists should experience a high index of suspicion.
Conclusion
This case reinforces the diagnostic complexities encountered during breast cancer management, emphasizing the need for a multidisciplinary approach and judicious use of diagnostic modalities to improve diagnosis precision and allow a tailored treatment plan for better patient prognosis and outcomes.
1. INTRODUCTION
Invasive cribriform breast cancer (ICC) is a rare type of carcinoma, constituting 0.3-3.6% of all primary invasive breast cancers. It is characterized by its distinctive cribriform structure, which resembles a small sieve-like appearance under microscopy, thus forming the key to microscopic diagnosis. ICC's unique morphology results from malignant cells forming well-defined lacunae similar to a fenestrated plate and exhibits a unique histological and clinical behavior [1].
The ICC is usually seen in older menopausal women, and it rarely affects males. It has the same risk factors for breast carcinoma, including positive family history and genetic factors, i.e., BRCA1 and BRCA2 gene mutations. Prolonged exposure to estrogen history in addition to obesity and sedentary life is associated with increased risks [2]. Affected cases present with breast mass or nipple discharge, or they are accidentally discovered during mammographic examinations. Histologically, ICCs are classified into pure, classical, and mixed ICCs [1, 2].
Pure ICC comprises exclusive cribriform cells and tends to have the best prognosis and least tendency to lymph node (LN) metastasis. The classical ICC is a mixture of cribriform and other types of breast cancers as papillary type. The mixed ICC includes cribriform ICC with other invasive cancers, like ductal carcinoma. This classification has prognostic and therapeutic implications for treatment plans [3]. Presently, there is more than one histopathological classification system for ICC. The WHO classification considers tumors showing more than 90% of a special tumor type as pure special type. Tumors that do not meet these criteria, including those with mixed patterns, are called invasive cancer, if not otherwise specified (NST). For that, the broader and less strict definition of pure ICC that aligns with classical ICC is no longer valid [4] (Table 1) [2, 5-7].
Other grading systems are also employed regardless of the cancer subtype. Those include the nuclear grading, which relies on the Nottingham grading system, and the TNM staging system, which assesses the tumor's overall extent [8].
ICC usually has a good prognosis compared to other invasive carcinomas, with its low tendency of lymph node (LN) metastasis and high rate of positive immunohistochemical markers, being positively linked with improved patient survival [9].
Here, we present a comprehensive analysis of the management of a 62 year-old lady with ICC of the breast. A delineation of the diagnostic and therapeutic steps undertaken is provided, critically discussing the challenges encountered and how they have been addressed to reach the underlying diagnosis. The current case underscores the importance of a holistic patient-centered approach to optimize patient outcomes.
2. CASE PRESENTATION
A 62 year-old lady presented with a history of right breast nipple discharge for a duration of 2 months. She mentioned an underlying mass in the same breast that was getting larger during the last six months; however, she did not seek medical evaluation until she observed a serosanguinous bloody discharge from the breast, which was not voluminous.
On examination, the right breast showed no signs of skin changes, nipple changes, or retraction. There was no discrepancy in size compared to the contralateral breast. Local examination revealed a 7x4.5 cm painless mass. It showed no fixation, neither to the skin nor to the underlying structure. Axilla examinations showed no palpable LN on either side. The patient was a multiparous woman and had experienced menopause since the age of 52 years. Her past medical and surgical history was unremarkable. History of drug allergy or smoking was negative; she did not report a prior history of oral contraceptive use.
Mammography was chosen as the primary investigation owing to its ability to assess suspicious breast masses and guide the decision for additional investigations if any malignancy is suspected. It revealed two adjacent oval-shaped hyper dense masses that were partially well-circumscribed, collectively measuring 6x4 cm in the retro-areolar region of the right breast, immediately posterior to the nipple region; a focal asymmetry was noticed in the post aspect of the lesion. The breast was rated BI-RADS 4 in accordance with the breast imaging reporting and data system, implying a definitive probability of a malignant pathology (Fig. 1).
Target ultrasound (US) was used next to evaluate the suspicious mass better, differentiate solid and cystic components, and assess the retro-areolar region, which is challenging in mammography. It showed two oval-shaped complex cystic lesions with internal echoes located posterior to the nipple region. The lesions had relatively thin wall lesions, and the largest measured about 2.3x1.6 cm with heterogeneous internal soft tissue components (Fig. 2A). Color Doppler US showed increased peripheral and internal vascularity, as depicted in Fig. (2B). This finding was highly suspicious of malignancy; given the diagnostic uncertainty in imaging studies, a tru-cut biopsy under US guidance and local anesthesia was recommended to have a deeper insight into the architectural details and enable immunohistochemistry examination. The differential diagnosis at this stage was encapsulated papillary carcinoma or cystic type of invasive breast cancer with tumor necrosis. The possibility of breast hematoma can be considered if there is a history of trauma [4].
A tru-cut biopsy revealed a tumor of papillary architecture lined with atypical ductal epithelial cells that showed nuclear atypia (Fig. 3); the pathologist gave two probable diagnoses, i.e., a papillary lesion with atypical hyperplasia or ductal carcinoma in situ, and advised for excisional biopsy for better assessment. The immunohistochemistry staining (IHC) was positive for the estrogen receptor ER (95% strong staining) and negative for P63, i.e., myoepithelial cells.
| ICC Type | Definition | Notes | Refs. |
|---|---|---|---|
| Pure ICC | Tumor showing more than 90% cribriform morphology | Described in both WHO 4th and 5th editions |
[5] |
| Classical ICC | Tumors showing mainly a cribriform pattern, in addition to less than 50% tubular CA | A term that was used previously, but is currently less relevant in the newer WHO edition | [2] |
| Mixed ICC | Tumors having cribriform patterns ranging from 50-90% and 10-49% of another CA component (non-tubular) | Recognized in WHO 4th edition | [6] |
| Invasive carcinoma, NST | Tumors missing a cribriform pattern being predominant or showing a special type pattern of less than 90% | Currently classified as invasive carcinoma, if not otherwise specified (NST) | [7] |
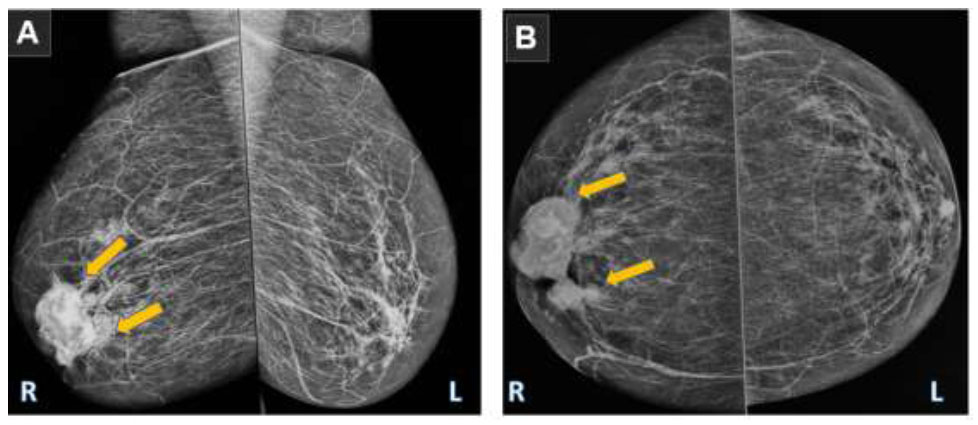
Digital mammography. (A) Mediolateral oblique (MLO) and craniocaudal (CC) views; (B) there were two adjacent oval-shaped hyper-dense partially well-circumscribed masses seen at the retro-areolar region, immediately posterior to the nipple region, with focal asymmetry at the post aspect of the lesion (orange arrows). This monograph was classified as BI-RADS 4, implying a definitive probability of malignancy, and the patient was referred for a tru-cut biopsy under US guidance.
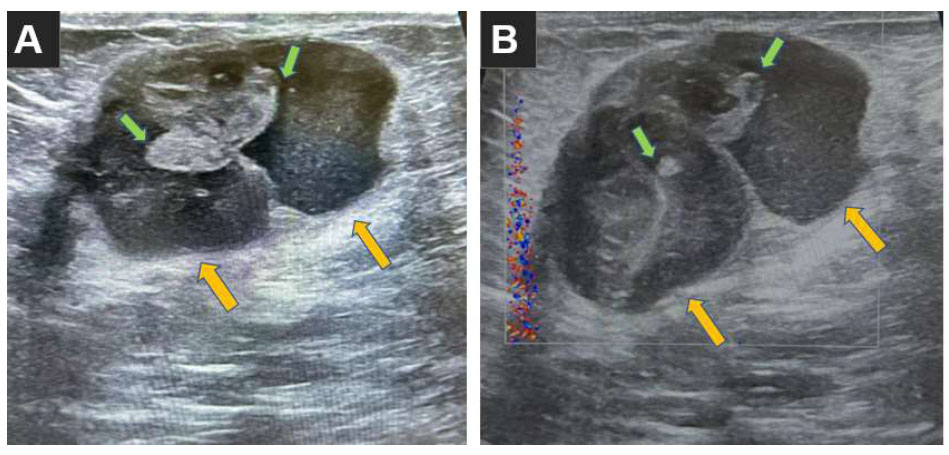
(A) Target ultrasound showing oval-shaped cystic lesions with internal echoes posterior to nipple region (orange arrows) of relatively thin wall, collectively measuring about 41 x 24 mm, with the largest one measuring about 23 x 16 mm, showing heterogeneous internal soft tissue component; green arrows indicate internal vascularity seen at color Doppler US. (B) Suspicion of malignancy, leading to the recommendation for biopsy under US guidance.
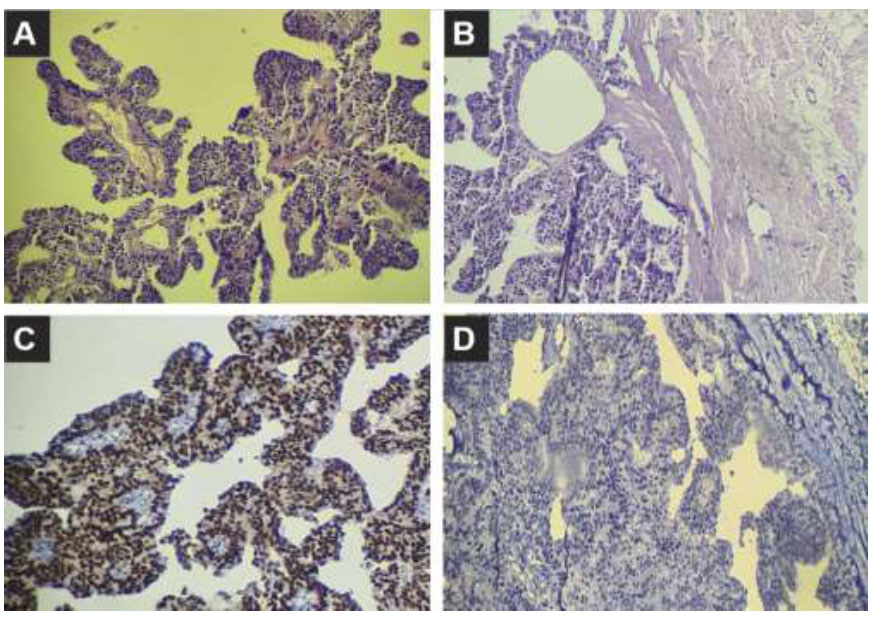
Tru-cut biopsy results. (A, B) Papillary architecture lesions. (C, D) Immunohistochemistry showing strong positive ER (C) and negative p63 (D).
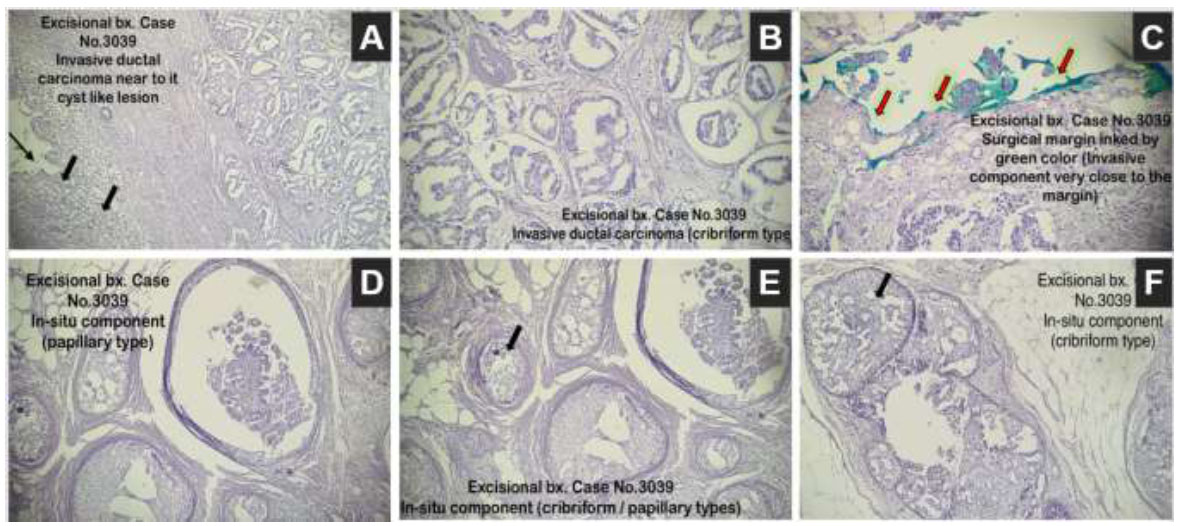
Excisional biopsy revealed a cribriform invasive ductal carcinoma (black arrows) (A, B). Positive margins (C), highlighted by red arrows. There were areas with ductal carcinoma in situ of both cribriform and papillary types (D-F).
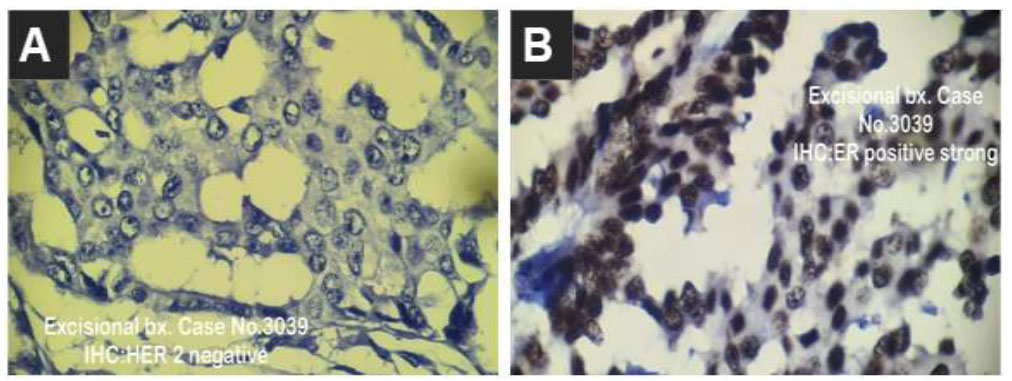
IHC for excisional biopsy showed negative HER 2 receptors (A) and strong positive estrogen receptors (B).
Given the advice of the pathologist and to have a complete view of the tumor and its surroundings, an excisional biopsy under general anesthesia was deemed necessary; the histopathological report showed an invasive cribriform ductal carcinoma grade I (according to Nottingham's modified scoring system), and the surrounding breast tissue showed proliferative fibrocystic breast changes, intraductal papilloma, and duct ectasia (Fig. 4). The specimen was negative for lymph-vascular invasion, and perineural invasion was seen. However, the tumor reached the surgical margin and was classified as TNM staging pT3. The IHC report was positive for ER and negative for PR and HER2 receptors (human epidermal growth factor receptor 2) (Fig. 5).
| Parameters |
1st Study (Demir et al., 2021 [4]) |
2nd Study (Mo et al., 2017 [2]) |
3rd Study (Cong et al., 2015 [8]) |
|---|---|---|---|
| Included cases | 42 cases | 12 cases | 9 cases |
| Sex | 96.5% female | 100% female | 100% female |
| Age range (years) | 50-79 | 38-75 | 40-79 |
| Tumor laterality site | Rt in 60.2% | Not documented | Rt 55.5% |
| Tumor size (cm) | <1 to >5 | 2 to 10 | 1.0 to 3.2 |
| Negative lymph node status | 50.2% | Not documented | Less than 2 in most cases |
| The irregular shape in the US | 91.5% | Not documented | 88.89% |
| Spiculated margin in the US | 70.35% | Not documented | 88.89% |
| Hypogenic in the US | 100% | Not documented | 100% |
| Positive acoustic shadow on US | 19.85% | Not documented | Not documented |
| Orientation (parallel) | 90.055 | Not documented | Not documented |
| Estrogen (E2) and progesterone (P) receptors | E2:100%; P: 96% | Moderately to strongly positive (30%-95%) | E2: 100%; P: 100% |
| HER2 status (negative) | 96% | 91.67% | 77.8% |
| Other markers | K˙I67 status ≤14% positive in 85.35% |
• p53 focal positive in 16.67% • CK5/6 negative • CD10, p63 focal positive in 8.33% |
Calponin, p63, CK5/6, CD10: negative |
| Surgical approach | Mastectomy in 40% and breast-conserving surgery (BCS) in 60% | Mastectomy in 83.33% and rest by breast-conserving surgery | Mastectomy in 44.4% and rest by breast-conserving surgery |
| Additional therapy | 100% hormonal | 66.65% chemotherapy | • 33.35% of the cases received chemotherapy • Radiotherapy was given to 33.35% of the cases • Hormonal therapy was given to 33.35% of the cases |
| Follow-up (months) | 15-106 | 12-70 | 4-70 |
3. DISCUSSION
This report discussed the diagnostic scheme for a 62-year-old postmenopausal woman who presented with a right breast nipple discharge and a palpable mass measuring 6x4 cm. The ultrasound showed complex cystic lesions with internal echoes and heterogeneous internal soft tissue components. The diagnosis of ICC was suspected by imaging (US and mammography) and was confirmed by histopathological examination with the key cribriform structure, supported by IHC.
A comprehensive summary of literature reviews published in the last 10 years was conducted, highlighting the demographic criteria, imaging modalities, surgical interventions, and IHC findings alongside the follow-up period, as outlined in Table 2.
Several challenges are associated with ICC diagnosis. First, ICC may present with subtle or atypical imaging criteria resembling benign pathologies, like sclerosing adenosis, leading to underdiagnosis or misclassification by imaging studies [7].
Second, ICC may regress, which may further add complexity to the detection and diagnosis [10]. Histologically speaking, ICC may be challenging for the pathologist due to overlapping features with other breast cancers [11]. Furthermore, ICC can coexist with other types of breast cancer, which urges careful evaluation to unveil its unique characteristics. ICC exhibits a diverse nucleus grading that is linked with its clinical course [12].
The reliance on IHC markers is not always definitive, as it may show borderline patterns that need meticulous pathological evaluations [13]. Finally, we have to acknowledge inter-observer variability among pathologists and radiologists [14], imposing an additional layer of diagnostic uncertainty when differentiating ICC from lower-grade carcinomas [15]. These limitations urge radiologists and pathologists to experience a high index of suspicion to improve diagnostic precision.
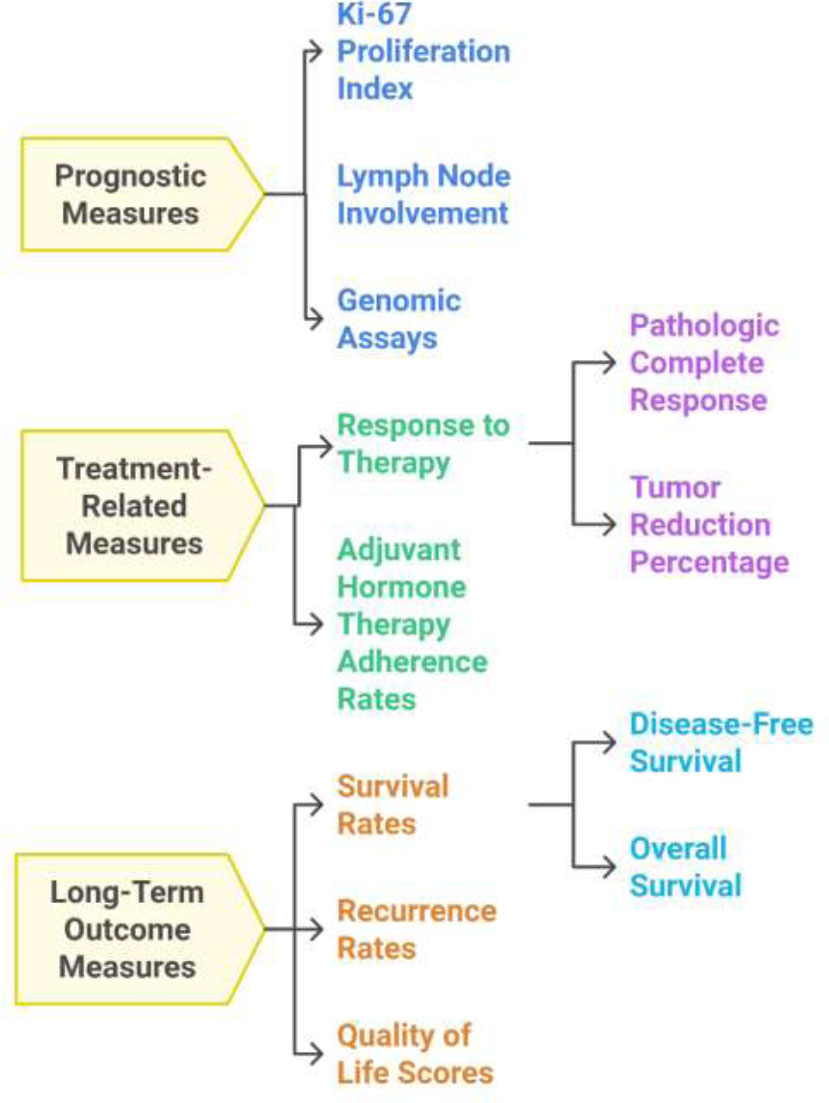
Important quantitative measures that are needed for a comprehensive approach to managing ICC of the breast, offering a tailored, patient-centered approach to improve the outcomes.
The current case was classified as BI-RADS 4, suggesting papillary or invasive carcinoma. While cytology proposed a ductal carcinoma in situ or papillary tumor, the histopathology and IHC unraveled ICC diagnosis. It is not unusual to have a discrepancy between US-ideated core needle biopsy and excisional biopsy. The former may fail to capture the ICC structure owing to sampling issues, leading to misdiagnosis. Excisional biopsies are more reliable for a definitive diagnosis supported by IHC [2].
The IHC stain plays a crucial role in ICC management. It confirms biological markers associated with ICC cribriform pattern, excludes other diagnoses, and helps exclude associated ductal carcinoma, which changes the patient staging and treatment plans. The presence of hormonal markers (estrogen receptor (ER) and progesterone receptor) improves patients' survival with hormone therapy [16]. Moreover, these markers provide a deeper insight into tumor behavior, progress, and LN involvement. For example, a lack of p63 expression can discriminate invasive tumors from in situ lesions. Thus, IHC markers allow for tailored patient follow-up plans [13].
We believe that the current study has potential implications for practice. It emphasizes the importance of a multidisciplinary approach in addition to the integration of advanced diagnosis modalities. This is critical, especially when facing subtle and overlapped findings in the clinical, imaging, and histopathological reports [1].
Incorporating advanced diagnostic modalities, such as digital tomosynthesis and MRI, alongside conventional mammography, is needed to improve diagnosis. Collaboration between radiologists and pathologists is important, especially when facing atypical presentations, in order to improve diagnosis and reduce cases of misclassification [17, 18].
Future research should prioritize identifying newer imaging modalities and leveraging molecular profiling to refine ICC diagnosis and classification [19]. Additionally, exploring the natural history of ICC progression may help improve its diagnosis [3]. The revolutionary potential of artificial intelligence in medicine may present a valuable tool for subtle breast imaging, offering more efficient diagnostic strategies [20-22].
Compared to other invasive breast carcinomas, ICC has a better prognosis, showing lower grades, smaller size, less tendency for LN metastasis, higher rates of hormonal expression, and lower HER2 amplification than invasive ductal carcinoma [23]. Liu et al. proposed a nomogram to standardize guidelines for ICC prognosis; they showed that patients' age, marital state, tumor locality, and IHC state (estrogen receptor), in addition to T and M staging, had a significant impact on ICC survival and prognosis [9]. The treatment for ICC is surgical, whether involving breast-conserving surgery or modifiable radical mastectomy with or without adjuvant chemoradiotherapy [24, 25].
Recently, there has been a shift to hormonal therapy based on the IHC results, including tamoxifen and letrozole. However, there are no fixed guidelines regarding this topic owing to the indolent nature of the tumor [1, 26]. Important quantitative metrics in the ICC of the breast include diagnostic, prognostic, treatment-related, and long outcome measures [27-29], all of which are summarized in Fig. (6).
CONCLUSION
This case report has provided deeper insights into the diagnostic dilemmas imposed by ICC. It has reinforced the importance of a high index of suspicion and integrating a multimodal diagnostic approach in breast tumors. A different potential diagnosis was made at each diagnostic step for this case, and the actual cause remained elusive until histopathology and IHC results were integrated. The current case has offered a practical framework for similar challenging cases. Future work is warranted to standardize diagnostic criteria and examine newer emerging modalities, such as artificial intelligence, to improve diagnosis and patient outcomes.
AUTHORS’ CONTRIBUTION
It is hereby acknowledged that all authors have accepted responsibility for the manuscript's content and consented to its submission. They have meticulously reviewed all results and unanimously approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| ICC | = Invasive cribriform cancer of the breast |
| LN | = Lymph node |
| NST | = Invasive cancer, if not otherwise specified |
| BI-RADS | = Breast imaging-reporting and data system |
| US | = Ultrasound |
| ER | = Estrogen receptor |
| IHC | = Immunohistochemistry staining |
| PR | = Progesterone receptors |
| HER2 | = Human epidermal growth factor receptor 2 |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The ethics committee of Mutsansiriyah University, College of Medicine, Iran, provided approval for the publication of this case report (IRB: 274; dated 12/12/2024).
HUMAN AND ANIMAL RIGHTS
All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
AVAILABILITY OF DATA AND MATERIALS
The data and supportive information are available within the article.
ACKNOWLEDGEMENTS
The authors would like to thank Al- Mustansiriyah University, Iran for its continuous support.


