All published articles of this journal are available on ScienceDirect.
Accuracy of Ultrasound in Bone Age Estimation in Iraqi Short-stature Children
Abstract
Background
Bone age assessment represents an important step in the management of children with Isolated Growth Hormone Deficiency (IGHD). This study examined the usefulness of Ultrasound (US) in the assessment of bone age in a sample of Iraqi children with IGHD as compared to radiography as a reference. Additionally, it verified if patient gender and growth hormone therapy have an impact on US accuracy.
Methods
An observational cross-sectional study recruited children with isolated growth hormone deficiency who were diagnosed and followed at the Alresafa Specialized Center for Endocrinology and Diabetes, Baghdad, Iraq, over 6 months. Children with IGHD from Iraqi nationality were recruited, while children from other nationalities or having multiple hormonal deficiencies, syndromic features, and parent refused participation were excluded. For each patient, a bone age assessment was conducted using two methods: US and TW2 hand-wrist radiographs at the same visit by the same radiologist.
Results
A total of 116 children were included. The chronological age of recruited children was 7 to 17 years, with a mean of 13.01 ± 2.78 years. There were 67 males (57.9%) with a male-to-female ratio of 1.37:1. The patient's gender did not affect the US accuracy; there was a non-significant difference in the bone age estimated by the US and conventional radiograph for both male and female patients, (p-value = 0.087, 0.308) respectively. Those who received growth hormone therapy and those who did not for both male and female patients (p-value = 0.071,0.243), respectively. There was a strong positive correlation between the means of bone age assessed by ultrasound (US) and conventional radiography for both males and females, with correlation coefficients of r = 0.788 and r = 0.703, respectively.
Conclusion
Ultrasound may serve as a valid replacement for radiography in the assessment of bone age in children with short stature caused by a growth hormone deficiency, irrespective of the gender and treatment received. Thus, it may overcome radiography drawbacks for children who need sequential bone age assessment.
1. INTRODUCTION
Short stature in children is a common parental concern, which is defined as having a height of more than 2 standard deviations and a score below the mean [1, 2]. Although it represents a prevalent manifestation of endocrine diseases in pediatrics, on evaluation, only a small percentage of children will be found to have a pathological short stature [3]. Growth hormone deficiency is one of the pathological causes of short stature, in which the diagnosis is suggested clinically and confirmed by growth hormone provocative tests [4]. Bone age assessment represents an important step in the evaluation of children with growth hormone deficiency [5].
For years, hand-wrist radiography was the gold standard tool used to assess bone age using different methods, including the Greulich and Pyle (GP) and Tanner-Whitehouse (TW) methods [6]. The GP method involves comparing the left hand and wrist radiographs with a set of reference images in a standard atlas. It is relatively straightforward and commonly used in clinical practice. On the other hand, the TW method is considered more precise and detailed as it involves a scoring system to assess each bone separately [7]. Advances in bone age assessment have resulted in the introduction of newer methods, including computerized automatic systems [8, 9] and magnetic imaging of the hand [10]. In addition, artificial intelligence models are being tested to overcome the long reading time and interobserver variability [11].
Physicians were anxious about the cumulative ionizing dose from the wrist X-ray during bone age assessment. As a result, ultrasonography gained an increasing interest in bone age assessment because of its reliability and safety [12]. Previous studies discussed the reliability of bone age assessment by ultrasound and confirmed that there is a considerable variation in bone age between ethnic groups and sex. In the presence of pathological conditions, this study was conducted to assess the usefulness of ultrasound to assess bone age in a sample of Iraqi children with isolated growth hormone deficiency as compared to radiography as a reference [6, 13].
2. METHOD
An observational cross-sectional study recruited children with isolated growth hormone deficiency who were diagnosed and followed at the Specialized Center for Endocrinology and Diabetes, Baghdad, Iraq, over a 6-month period from 1st of April 2024 to 30th of September 2024. Patients were recruited after matching the following inclusion criteria: (1) Iraqi children with parental concern of short stature (2) the cause of short stature is isolated growth hormone deficiency confirmed by GH provocative test.
Children were excluded from the study if they matched any of the following exclusion criteria: (1) Age > 18 years, (2) children with non-Iraqi nationality (3), multiple pituitary hormone deficiency, (4) children with other causes of short stature 5) children with syndromes or dysmorphic features, and (5) parents of children who refuse to participate in the study. The study was approved by the scientific ethical committee of AL Kindy College, Baghdad University.
For each patient, a bone age assessment was conducted by two methods (ultrasound and TW2 hand wrist radiographs) at the same visit and by the same radiologist to decrease the chance of bias and avoid observers' variability.
The ultrasound method of assessing the bone age of the included children was performed in the radiology department by two specialist radiologists who had (5-10 years)' of experience in MSK ultrasound, especially bone age evaluation. The children's age and other medical details were concealed from the radiologists while they scanned and measured them in the US, and both examined the children blindly to avoid bias. GE LOGIC S8 ultrasound machine was used to conduct hand-held ultrasound using a 9 MHz linear array transducer. Two sites were examined by ultrasound: the LT wrist joint (distal radial and distal ulnar bones) and the LT knee joint (distal femoral bone). All the included members did an LT wrist X-ray during the same visit for an ultrasound exam.
The method of the ultrasound examination was explained to the child and his parents before the exam. The ultrasonic probe was positioned longitudinally along the distal lateral aspect of the forearm in the sagittal plane for imaging the radius. Pronation of the forearm was used to image the styloid process while the probe was positioned longitudinally along the distal lateral aspect in the coronal plane for ulna imaging. Finally, for imaging the femoral epicondyle, the probe was placed along the medial aspect of the knee joint in the coronal plane (Fig. 1). The ratio of the maximum height of the echogenic ossification center to the whole epiphysis, including the hypoechoic cartilaginous part, was measured for the three mentioned bones. The three values were then summed and multiplied by 100 to obtain the ultrasonic skeletal maturity score. The ultrasonic bone age was obtained using a list of typical age-appropriate score values. The age of boys and girls whose scores fall within the 50th percentile [14] was then determined [14].
Bone age assessment by conventional hand-wrist radiography was based on the TW-2 RUS (radio, ulna, and selected metacarpals and phalanges) method, which involves a scoring system. It examines the development of 20 bones, hand and wrist bones, and their ossification. Each of these bones is assessed for the developmental stage, which is assigned to a certain score, and the sum of the total score is compared to a reference that identifies a certain bone age [15, 16].
The sample size calculated by the following formula is [17]:
N = (Za/2 + Zb) 2 / r2
where, r = 0.26 (expected correlation) taken from earlier studies [18], a = 0.05, which is the significance level, power = 0.8 (desired power), Za = Standard normal variate for level of significance, and Zb = Standard normal variate for power or type 2 error.
N = ((Za/2 + Zb)2 / r2)
N = ((1.96 + 0.84)2 / 0.262) N ≈ 116
Therefore, the required sample size is 116.
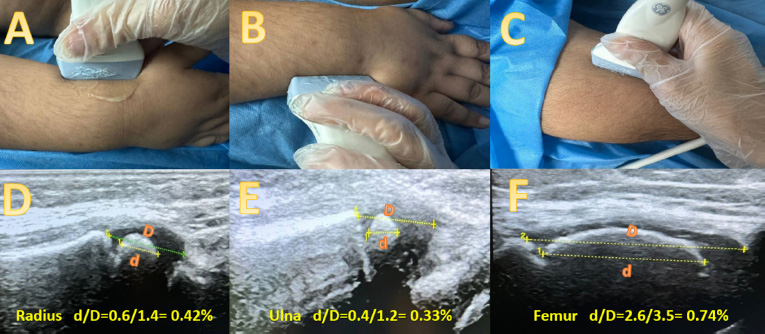
Ahows the US examination of the left wrist and left knee joints. A-C photos show the probe positioning during exam A) at the distal radius, B) at the distal ulna and C) at the distal femur. D-F shows the way by which we measured the ossification centers. (the orange d) represents the echogenic part of the epiphysis measured as the greatest height of the ossification center, while (the orange D) represents the hypoechoic epiphysis, including the echogenic ossification center. The d/D represents the ratio.
2.1. Statistical Analysis
SPSS version 25 was used to conduct data analyses. After ensuring the data was normally distributed by Kolmogorov–Smirnov and Shapiro–Wilk tests, the ANOVA test was used to determine the significance of the difference among more than two means when comparing quantitative data, and the student's t-test was used to compare two independent means. ANOVA is a statistical technique that compares the means of multiple groups at the same time to ascertain whether observed differences are the result of chance or represent true differences. Multiple factors and their interactions can be handled by ANOVA, which offers a reliable method for comprehending complex relationships [19]. Pearson correlation was used to test the significance between two quantitative variables. A p-value level of less than 0.05 was taken as significant.
3. RESULTS
A total of 116 children with isolated growth hormone deficiency were included. The participants ranged in age from 7 to 17 years, with an average age of 13.01 ± 2.78 years. In terms of gender distribution (following SAGER guidelines), there were 67 males (57.9%) and 49 females (42.1%), resulting in a male-to-female ratio of 1.37:1, as illustrated in Fig. (2).
The mean chronological age of male children was 13.18 ± 2.03 years, while the means of the conventional radiographic and ultrasonographic bone ages were 12.52 ± 1.85 and 12.63 ± 1.50 years, respectively. There was no statistically significant difference between the means of chronological age, conventional radiographic, and ultrasonographic bone ages (p= 0.087). In the females, the means of chronological age, conventional radiographic, and ultrasonographic bone ages were 11.43 ± 2.91, 11.10 ± 2.05, and 10.75 ± 1.21, respectively. As in male patients, there was no significant difference in the means of chronological age, conventional radiographic, and ultrasonographic bone ages of the females (p= 0.308), as illustrated in Table 1.
Gender distribution within the study group.
“Regarding growth hormone therapy, about two-thirds of the children were on treatment (76, 65.8%), while the remaining 40 (34.2%) did not receive treatment. Among children who had growth hormone therapy, there was no significant difference among the means of chronological age, conventional radiographic, and ultrasonographic bone ages (10.84 ± 2.98 vs 10.15 ± 2.03 and 10.81 ± 2.05, p= 0.243).
| Patients Gender | Evaluation |
Bone Age Mean ± SD |
p-value* |
|---|---|---|---|
| Male | Chronologic | 13.18 ± 2.03 | 0.087 |
| Radiographs | 12.52 ± 1.85 | ||
| Ultrasonography | 12.63 ± 1.50 | ||
| - | |||
| Female | Chronologic | 11.43 ± 2.91 | 0.308 |
| Radiographs | 11.11 ± 2.05 | ||
| Ultrasonography | 10.75 ± 1.21 | ||
| Growth Hormone Therapy | Evaluation |
Bone Age (years) Mean ± SD |
p-value* |
|---|---|---|---|
| NO | Chronologic | 13.38 ± 2.20 | 0.071 |
| Radiographs | 11.68 ± 2.17 | ||
| Ultrasonography | 11.73 ± 1.83 | ||
| - | |||
| YES | Chronologic | 10.84 ± 2.98 | 0.243 |
| Radiographs | 10.15 ± 2.03 | ||
| Ultrasonography | 10.81 ± 2.05 | ||
| Gender | Evaluation of Bone Age | Radiography | Ultrasonography | ||
|---|---|---|---|---|---|
| r | p-value | r | p-value | ||
| Male | Chronological Age | 0.864 | 0.001 | 0.837 | 0.001 |
| Ultrasonography | 0.788 | 0.001 | – | – | |
| - | |||||
| Female | Chronological Age | 0.836 | 0.001 | 0.753 | 0.001 |
| Ultrasonography | 0.703 | 0.001 | – | – | |
At the same time, the means of chronological age, conventional radiographic, and ultrasonographic bone ages were not significantly different in the children who didn not receive hormonal therapy (13.38 ± 2.20 vs. 11.68 ± 2.17 and 11.73 ± 1.83, p= 0.071), as illustrated in Table 2.
For the males, the Pearson correlation analysis showed a significant positive correlation between chronological age and radiologic bone age (r= 0.864, p= 0.001) and ultrasonic bone age (r= 0.837, p= 0.001). Further, there was a strong positive correlation between radiologic and ultrasonic bone age (r= 0.788, p= 0.001). The same findings were observed among female children where chronological age was significantly correlated with radiologic bone age (r= 0.836, p= 0.001) and ultrasonic bone age (r= 0.753, p= 0.001), and a strong correlation was detected between radiologic and ultrasonic bone ages (r= 0.703, p= 0.001), as shown in Table 3.
4. DISCUSSION
This study compared the bone age estimated by ultrasound to those estimated by conventional radiography in a sample of short-stature Iraqi children with growth hormone deficiency and demonstrated a non-significant difference between the means of bone age assessed by ultrasound and conventional radiographic method regardless of the gender of the children and growth hormone treatment. There was a strong correlation between the means of bone age assessed by ultrasound and conventional radiography. So, ultrasound may be considered a valid replacement for radiography in the assessment of bone age in children with short stature caused by growth hormone deficiency, as those children receive sequential bone age assessment to overcome the drawbacks of radiography [6, 20, 21].
Bone age represents skeletal maturation in the pediatric age group, and it has a wide spectrum of implications in pediatric endocrinology, like estimation of expected final adult height and diagnosis and management of various pediatric health conditions, including children with growth hormone deficiency [22, 23]. Estimation of bone age in children with short stature from growth hormone deficiency is conductedinitially to confirm the underlying cause and in following up patients with hormonal replacement therapy [24]. Children with growth hormone therapy had annual bone age estimation, and with the use of the conventional radiographic method, this will place the child at risk of a cumulative dose of ionizing radiation [25].
Multiple methods have been used in practice to assess bone age from wrist-hand X-rays, including the GP and TW methods, and each of these methods has its pros and cons [6]. The TW2 method used in the current study is more precise. It has much less inter and intra-observer variability in bone age assessment but is more time-consuming than the GP method, and since it also depends on wrist-x ray so has ionizing radiation risk [16]. As a result, ultrasound appears to be a more attractive method for bone age estimation as all drawbacks of radiology can be overcome with ultrasound use. Additionally, it is of low-cost, is readily available, and is non-ionizing [26].
Ultrasound is infrequently used for bone age estimation in routine practice due to the lack of training needed for its estimation and the limited availability of qualified US devices. For that, radiography is more prevalent since it is validated and accessible [27]. Moreover, studies addressing US accuracy in bone age determination remain scarce. Most recent research is currently focusing on newer emerging technologies, such as three-dimensional US, which has improved diagnostic precision, especially when combined with AI technology [28].
In the current study, there were non-significant differences in bone age estimated by ultrasound and conventional radiographs for both male and female patients with growth hormone deficiency (p-value = 0.087, 0.308), respectively.
Traditional radiology methods use gender-specific adjustment owing to different maturation rates for different genders, especially in terms of puberty [29].
In contrast, US scanning of bone age showed comparable accuracy regardless of gender. This advantage for the US can simplify bone age assessment and reduce the dependence on individualized calibration. This is valuable for pediatric endocrinology, where reducing radiation exposure is desired [8].
A study by Pin Lv showed good accuracy for both males and females by US examination; however, there was a higher accuracy for females owing to the favorable effect of estrogen on bone growth [30, 31].
The estimation of bone age was not affected by whether the patient started growth hormone therapy or not, as there were non-significant statistical differences in bone age estimated by conventional radiography and ultrasound between patients who received growth hormone therapy and those who did not start the hormonal therapy for both male and female patients (p-value = 0.071,0.243), respectively.
In the present study, the bone age assessed by ultrasound strongly correlated with that assessed by the TW2 method based on wrist x-rays for both males and females with r =0.788 and 0.703, respectively, and the correlation was statistically significant with p-value =0.001 for both. This means that regardless of the gender of the child, ultrasound can be used to assess bone age in children with short stature. This disagrees with the result of the study by Khan et al. on 100 children, which did not recommend the use of ultrasound in bone age assessment, as they found a poor correlation between bone age assessed by ultrasound and the conventional radiographic method [32]. This difference could stem from multiple reasons: first, the demographic characteristics of children in each study were different. Second, in the present study, only children with growth hormone deficiency were included, while in the Khan et al. study, children with short stature, irrespective of the cause, were included. Recently, artificial intelligence has emerged as a promising tool in the estimation of bone age in children presented with short stature, offering enhanced accuracy, consistency, and efficiency compared to traditional methods. AI algorithms, particularly those leveraging deep learning and convolutional neural networks, can analyze radiographic images to provide precise bone age assessments, reducing inter-observer variability and saving time for pediatricians [33-35].
This study had multiple limitations: First, the limited number of participants and the single center setting, which limited the generalizability of the results. Second, the radiological assessment of bone age was performed using the TW2 method as this is still the main method for bone age assessment used by pediatric endocrinologists in Iraq compared to TW3, commonly used in more developed countries. This method had multiple drawbacks, including outdated reference data, limited scope, less precision, and being subjected to less updated research, which makes it less aligned with the most recent updated findings [7, 15, 36, 37]. Third: the evaluation of bone age by ultrasound (US) requires the patient's presence for examination, followed by measuring the result and recording it on the skeletal maturity chart. In contrast, bone age assessment by X-ray is faster, easier, and does not require the patient's presence. Additionally, the X-ray film can be kept for comparison, whereas ultrasound is mainly operator-dependent, and the ultrasound images require several steps to assess the age for comparison [24].”Despite these limitations, this study represents the first paper that discusses the use of ultrasound for bone age estimation in children with growth hormone deficiency. It can lead to a large-scale multicentric study to confirm the results of the present study. In addition, it highlights the need to establish a bone age reference for Iraqi or at least Arabic children with the same ethnicity as a noticeable distinction amongst populations in the distribution of TW standard maturity scores in relation to chronological age. These variations may stem from ethnic diversity or from changes in biological maturity that occurred during the last century due to external influences, including lifestyle changes and socioeconomic advancements [38].
CONCLUSION
In children with growth hormone deficiency, ultrasound estimation of bone age strongly correlates with hand-wrist x-ray based bone age estimation regardless of gender and hormonal therapy, which holds significance for overcoming the drawbacks of conventional radiographic methods. Based on the current findings, the use of ultrasound represents a promising option for the initial diagnosis and follow-up of children with isolated growth hormone deficiency to ensure effective management. As such, patients may require repeated follow-up to assess the response to treatment, so frequent radiation exposure may be required.
AUTHORS’ CONTRIBUTION
M.M.H., S.K.A.: Data collection; Q.A.H.: Analysis and interpretation of results; N.N.A., W.N.M.H.: Draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| IGHD | = Isolated Growth Hormone Deficienc |
| GP | = Greulich and Pyle |
| TW | = Tanner-Whitehouse |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the ethics committee of the Al-Kindy Teaching Hospital in Baghdad, Iraq, ethical approval number is (217) on 27/5/2024.
HUMAN AND ANIMAL RIGHTS
All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
AVAILABILITY OF DATA AND MATERIALS
All the data and supporting information is provided within the article.
ACKNOWLEDGEMENTS
Declared None.


