All published articles of this journal are available on ScienceDirect.
Paroxysmal Lower Limb Tremor as a Rare Presentation of Colloid Cyst of the Third Ventricle: A Case Report and Literature Review
Abstract
Objective:
Colloid cysts of the third ventricle are benign intracranial tumors. They are most commonly presented with signs of intracranial hypertension due to obstructive hydrocephalus, including headache, nausea/vomiting and vision disturbances, whereas extrapyramidal symptoms such as tremor are very rare. Sudden death, due to abruptly developed hydrocephalus, can be also observed. Although paroxysmal symptomatology attributed to the intermittent obstruction of the foramen of Monro is considered the “classical” clinical presentation, it is rather the exception in clinical practice.
Case Report:
A 42-year-old woman with no medical history was admitted to the neurology department as suffering from a potential demyelinating disease due to episodes of paroxysmal tremor of her right lower limb and persistent mild gait instability, which presented 15 days prior to her arrival. She also complained of episodes of partially position-dependent bilateral headache over the last 10 years, as well as episodes of vertigo over the last 4 years. On arrival, her gait was shuffling and mildly wide-based and an intermittent tremor of her right lower limb was observed in supine and sitting positions, but not in a prone position. Brain magnetic resonance imaging (MRI) demonstrated a round cystic lesion of the third ventricle, accompanied by hydrocephalus with enlargement of lateral ventricles. MRI findings were highly indicative of a colloid cyst. The patient underwent resection of the mass and the tremor resolved after surgery.
Conclusion:
Given the greatly heterogeneous clinical presentation of colloid cysts, our case highlights the significance of the prompt diagnosis of this rare but potentially fatal cause of paroxysmal limb tremor.
1. INTRODUCTION
Colloid cysts are rare benign intracranial lesions derived from endoderm or primitive neuroepithelium [1], most commonly located on the roof of the third ventricle, adjacent to the foramen of Monro via which the third ventricle communicates with the lateral ventricles [2]. Although colloid cysts grow slowly and are usually asymptomatic, some patients present with heterogeneous and non-specific neurological complaints related to increased intracranial pressure due to obstructive hydrocephalus or compression of surrounding brain regions [3]. Most common clinical manifestations involve paroxysmal headache, diplopia, nausea/vomiting, loss of consciousness, gait impairment, cognitive deficits and lower limb weakness. Extrapyramidal symptoms, involving tremor, represent rare presenting complaints [4-7]. Although very rare in clinical practice, displacement of the cyst may obstruct the foramen of Monro, resulting in characteristic paroxysmal position-dependent symptomatology [6, 8]. Importantly, sudden death may occur, although rarely, due to acute hydrocephalus and subsequent herniation. Less frequently, cardiovascular complications may be encountered due to abrupt hypothalamic dysfunction [3]. Therefore, given the fact that colloid cysts are surgically treatable, a high level of suspicion is required and physicians should be aware of rare presenting symptoms in order to promptly diagnose this life-threatening clinical entity.
Herein, we present a case of a 42-year-old woman who was admitted to the emergency department complaining of paroxysmal tremor of her right lower limb and mild gait instability, which presented 15 days prior to admittance, finally attributing to the colloid cyst of the third ventricle. Symptoms were resolved upon surgical treatment. Furthermore, we provide a brief review of the literature in regard to epidemiology, clinical presentation, neuroimaging characteristics, treatment and prognostic factors of this rare but potentially fatal condition.
2. CASE REPORT
A 42-year-old woman was admitted to the neurology department with a clinical suspicion of a demyelinating disease due to paroxysmal tremor of her lower right limb and persistent mild gait instability over the past 15 days. Τhe episodes οf tremor of her lower right limb lasted about 5-15 seconds, occurred many times per day and did not depend on any specific position. She also complained of episodes of bilateral non-pulsing pressure-like headache in the frontal, parietal, and occipital region, with a frequency of about one or two times per week and short duration (from minutes to 3-4 hours), without nausea or vomiting over the past 10 years. Characteristically, the intensity of her headache episodes was at least partially dependent on her head and body position, since they worsened in the supine position and mildly improved when lying on her left or particularly right side. She also mentioned occasional episodes of vertigo with nausea, worsened by head movement over the past 4 years. Despite the history of recurrent headaches and vertigo, she never had neuroimaging before. There was no trauma history, complaint of visual impairment or episodes of loss of consciousness and the patient was otherwise in good health. Her past medical and family history was unremarkable. Upon arrival, her vital signs, mental status and general physical examination were normal. On neurological examination, pupils were round and reactive to light, there were no peripheral vision deficits and the examination of cranial nerves II-XII was also normal. Her gait was shuffling and mildly wide-based, deep tendon reflexes were more brisk in her right extremities, muscle strength of upper and lower limbs was 5/5 at MRC scale, and there was bilateral non-sustained ankle clonus with flexion plantar reflexes. An intermittent tremor of her right lower limb was observed, occurring in the supine and sitting, but not in the prone position. No sensation deficits, cerebellar or other extrapyramidal signs were identified. Electroencephalogram (EEG) did not show any clinically significant findings.
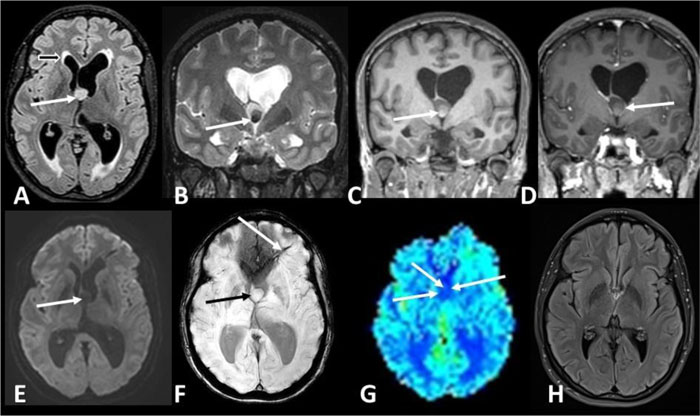
Magnetic Resonance Imaging (MRI) of the brain demonstrated a 1.5x1.2x1.1 cm round cystic lesion of the third ventricle, at the foramen of Monro, accompanied by obstructive hydrocephalus with enlargement of both lateral ventricles (Left>Right), whereas 3rd and 4th ventricles appeared normal. There was also high-intensity signal on T2-weighted images in the periventricular white matter, which is a sign of periventricular interstitial edema, secondary to transependymal resorption of cerebrospinal fluid (CSF).
The central and lower parts of the lesion had low signal intensity both on T1 and T2 sequences. The upper half and the periphery of the mass were hypointense on T1 and hyperintense on T2 images, without signal drop on fluid attenuated inversion recovery (FLAIR) images. Thin rim of peripheral enhancement post contrast medium administration was attributed to stretched septal veins, thus there was no enhancement of the lesion itself. No diffusion restriction was revealed on Diffusion-Weighted Imaging (DWI). Neither microbleeds nor calcifications were noticed on Susceptibility Weighted Imaging (SWI). Dynamic susceptibility contrast-enhanced perfusion imaging (DSC –PWI) revealed low values of relative Cerebral Blood Volume (rCBV) and relative Cerebral Blood Flow (rCBF). These findings were highly indicative of a colloid cyst of the third ventricle, resulting in obstructive hydrocephalus (Figs. 1A-1G). An incidental finding, a developmental venous anomaly in the left frontal lobe, extending from the left lateral ventricle to the left middle cerebral gyrus, was considered to be not clinically significant.
The patient was admitted to the neurosurgical department and underwent surgical resection of the mass without any peri- or post-operative complications. Histological examination of the lesion was consistent with a colloid cyst. The tremor of her right lower limb and gait instability resolved after surgery. At one-year follow-up, brain MRI demonstrated normal appearance of the lateral ventricles. No transependymal edema was observed (Fig. 1H).
3. DISCUSSION
Herein, we presented a case of a 42-year-old woman complaining of paroxysmal tremor of her right lower limb and mild gait instability, attributed to the colloid cyst of the third ventricle based on detailed neuroimaging testing, which was further confirmed histologically, whereas relevant images were not available for demonstration. In the following sections, we provide a brief review of the literature in regard to epidemiology, clinical presentation focusing on tremor, neuroimaging characteristics, treatment and prognostic factors of this rare but potentially fatal condition.
3.1. Epidemiology
It has been estimated that the incidence of colloid cysts is approximately 3.2/1.000.000 per year, representing about 0.5-2% of all intracranial neoplasms [2] and 15-20% of intraventricular tumors [7]. The onset age of clinical symptoms ranges from 2 months to 82 years and the mean age is about 30-45 years [5-9]. Some studies have indicated a slight male predominance [10-12], whereas others did not detect such differences [5, 9]. Although most cases of colloid cysts are sporadic, there have been described at least 10 familial cases in the literature, considered to be inherited in an autosomal-dominant manner [13].
3.2. Clinical Presentation
The rapidly expanding use and improved diagnostic accuracy of CT and MRI imaging have resulted in increased frequency of incidental colloid cysts [10], currently accounting for about 60% of cases [4]. When symptomatic, headache, nausea/vomiting, vision impairment, dizziness, gait instability, lower limb weakness, syncope and seizures are among the most common complaints [4-7]. Other symptoms involve progressive and/or fluctuating dementia, psychiatric disturbances, tinnitus and urinary or rectal incontinence [7]. Headache has been shown to be present in 90-95% of symptomatic cases [4], whereas hydrocephalus has been shown in about 50% of patients with symptomatic cysts [4]. The symptom duration ranges from a few hours to some years prior to diagnosis [8]. Headache episodes may be severe, paroxysmal, with short duration and more commonly located in the frontal region [7]. They may be also accompanied by loss of consciousness or drop attacks [7]. On neurological examination, papilledema, nystagmus, cerebellar signs and Babinski sign can be detected, although in several cases, the neurological examination is normal [2, 7].
Regarding tremor, literature search in MEDLINE database using the keywords “colloid cyst”, “clinical presentation”, “symptom”, “tremor”, and “extrapyramidal” in various combinations, as well as the additional search of the literature of each relevant article revealed 8 cases of colloid cysts of the third ventricle with tremor as a potential accompanying symptom or sign. As summarized in Table 1, Case 1 displayed a cerebellar tremor elicited in finger-to-nose and heal-to-toe tests [14]; in Case 2, patient’s tremor episodes were generalized like convulsions and followed by confusion while no tremor was observed in examination [14]; in Case 3, a generalized tremor throughout the body of the patient was accompanied by headache episodes with nausea and vomiting with no tremor mentioned in neurological examination [14]; whereas in Case 4, the fine tremor of the fingers attributed to chronic alcoholism [14]. In the 5th Case, headache, ataxic gait and tremor were the presenting symptoms, however, no more evidence was found for tremor [15]. Cases 6 and 7 were retrieved from a recent case-series study reporting 2 out of 24 cases of colloid cysts exhibiting tremor [16], while Case 8 refers to a patient with extrapyramidal symptoms [17].
| Case | Authors, Year of Publication | Type of Study | Description of Cases | Reference |
|---|---|---|---|---|
| 1st | Poppen J. L. et al., 1953 | Case series | 33-year-old man; presenting symptom: headache, gait unsteadiness and dizziness; neurological examination: right Babinski sign, right finger-to-nose tremor accompanied by right dysdiadochokinesia, and a bilateral coarse heel-to-knee test tremor accompanied by dysmetria, marked on the right | [14] |
| 2nd | Poppen J. L. et al., 1953 | Case series | 40-year-old man; presenting symptom: headache, increased appetite, vertigo, episodes of “generalized tremors, like convulsions,” followed by confusion; neurological examination: VI nerve weakness, nystagmus, non-sustained bilateral ankle clonus, dysmetria | [14] |
| 3rd | Poppen J. L. et al., 1953 | Case series | 22-year-old woman; presenting symptom: headaches accompanied by generalized tremor throughout her body and vomiting, dizziness, blurring of vision; neurological examination: no tremor mentioned | [14] |
| 4th | Poppen J. L. et al., 1953 | Case series | 42-year-old man; presenting symptom: headache, gait instability, drop attacks neurological examination: fine, rapid tremor of the fingers attributed to chronic alcoholism | [14] |
| 5th | Kimura H. et al., 1988 | Case report | 46-year-old woman; presenting symptom: headache, ataxic gait and tremor, no more evidence about tremor mentioned | [15] |
| 6th and 7th | Yurt A. et al., 2018 | Case series | 24 cases, mean age 41 years, neurological examination revealed tremor in 2 (8.33%) of the cases; no more evidence about tremor mentioned | [16] |
| 8th | Symss N. P. et al., 2014 | Case series | 80 cases, 1 patient reported extrapyramidal symptoms with no more relevant evidence | [17] |
Despite the fact that obstructive hydrocephalus has been reported to be associated with tremor, only occasionally it is the predominant symptom [18]. Furthermore, other movement disorders, such as chorea, parkinsonism and bobble head doll syndrome have been also reported in cases with obstructive hydrocephalus [19], but they are very rare particularly in cases of patients with colloid cysts. The exact pathophysiological mechanism underlying the colloid cyst-associated tremor is unclear. Neuroanatomically, given the fact that basal ganglia and thalamus are located under the ventricles, mechanical pressure on the cortico-striato-thalamic circuits from enlarged ventricles could only rarely result in the development of various types of movement disorders involving tremor, as presented in our case [18]. Of note, the tremor of our patient resolved completely after surgery, suggesting an important component of direct and reversible compression of basal ganglia pathways [20].
Therefore, given the non-specific and heterogeneous clinical presentation of the colloid cyst of the third ventricle, a high level of suspicion is required for prompt diagnosis, especially in young patients where only transient abnormal neurological signs are identified.
3.3. Neuroimaging
The size of colloid cysts ranges from 1-2 cm in diameter, whereas giant cysts (>3cm) have been also described [3]. On computed tomography (CT), colloid cysts appear as a round or ovoid lesion in the third ventricle, usually hyperdense, although iso- or hypodense colloid cysts can be also seen [8]. Rarely, calcifications and enhancement upon contrast administration can be detected [8]. The signal intensity of the colloid cyst on MRI depends on its protein and cholesterol content; most commonly, the lesion appears hypointense on T2-weighted and iso- or hyperintense on T1-weighted images [10]. Its signal intensity is associated with the viscosity of the fluid inside the cyst, which has been indicated to be helpful for stereotactic aspiration decision making; hyperdense colloid cysts are more difficult to aspirate in comparison to iso- or hypodense ones due to higher viscosity [10].
Colloid cysts are the most common masses of the third ventricles [7]. Differential diagnosis includes xanthogranulomas, hamartomas, pituitary adenomas and aneurysm of the basilar artery among others [21].
3.4. Prognosis and Treatment
It has been indicated that the risk of sudden clinical deterioration and death ranges from 3 to 12% for symptomatic colloid cysts [4, 6, 8], whereas the overall mortality rate is about 1.2% [22]. Headache or neuronal compression are indications for surgical intervention due to increased risk of hydrocephalus, rupture and chemical meningitis [23]. While surgical intervention is usually indicated for symptomatic cysts, the management of relatively small asymptomatic cysts depends on several characteristics and is still a debatable issue. Although it is rather unreliable to specifically define the natural history of colloid cysts owing to their low incidence in the general population, younger age at presentation, increased cyst volume, presence of headache, hyperintensity on FLAIR MRI imaging and the location of the cyst at the anterior or posterior extreme of the third ventricle have been associated with symptomatic cysts and obstructive hydrocephalus [4, 6]. Of note, these parameters were recently combined into a 5-point scale termed as Colloid Cyst Risk Score (CCRS), which well correlated with symptomatic cysts and the risk of hydrocephalus in a series of 163 patients [4]. This score was recently independently validated for both symptomatic and hydrocephalus-associated cysts, supporting its use in risk stratification of patients with colloid cysts, and clinical decision making [24].
There are several techniques that have been successfully employed in the treatment of colloid cysts, including transcortical-transventricular or transcallosal interforniceal microsurgical resection, endoscopic removal, stereotactic aspiration and simple CSF shunting [2, 17]. The most common peri- and post-operative complications of all surgical techniques include subdural and intracerebral hematoma, infection, seizures, venous infarction and memory deficits [2, 17]. In this context, a meta-analysis demonstrated that the microsurgical approach led to higher rates of full resection and reduced recurrence rates but increased morbidity rates in comparison to endoscopic removal [25]. However, there is still no consensus regarding the optimal treatment strategy.
CONCLUSION
Our case highlights the significance of prompt detection and subsequent early treatment of this rare but life-threatening cause of paroxysmal partially position-dependent limb tremor and instability. Given the greatly heterogeneous clinical presentation, a high level of suspicion is required for early diagnosis of symptomatic colloid cyst of the third ventricle, in order to avoid obstructive hydrocephalus and sudden potentially fatal clinical deterioration.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
Not Applicable.
CONSENT FOR PUBLICATION
Written informed consent was obtained from the patient.
STANDARDS OF REPORTING
CARE guidelines have been followed.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


