All published articles of this journal are available on ScienceDirect.
Prefrontal Brain Activation During Emotional Processing: A Functional Near Infrared Spectroscopy Study (fNIRS)
Abstract
The limbic system and especially the amygdala have been identified as key structures in emotion induction and regulation. Recently research has additionally focused on the influence of prefrontal areas on emotion processing in the limbic system and the amygdala. Results from fMRI studies indicate that the prefrontal cortex (PFC) is involved not only in emotion induction but also in emotion regulation. However, studies using fNIRS only report prefrontal brain activation during emotion induction. So far it lacks the attempt to compare emotion induction and emotion regulation with regard to prefrontal activation measured with fNIRS, to exclude the possibility that the reported prefrontal brain activation in fNIRS studies are mainly caused by automatic emotion regulation processes. Therefore this work tried to distinguish emotion induction from regulation via fNIRS of the prefrontal cortex. 20 healthy women viewed neutral pictures as a baseline condition, fearful pictures as induction condition and reappraised fearful pictures as regulation condition in randomized order. As predicted, the view-fearful condition led to higher arousal ratings than the view-neutral condition with the reappraise-fearful condition in between. For the fNIRS results the induction condition showed an activation of the bilateral PFC compared to the baseline condition (viewing neutral). The regulation condition showed an activation only of the left PFC compared to the baseline condition, although the direct comparison between induction and regulation condition revealed no significant difference in brain activation. Therefore our study underscores the results of previous fNIRS studies showing prefrontal brain activation during emotion induction and rejects the hypothesis that this prefrontal brain activation might only be a result of automatic emotion regulation processes.
INTRODUCTION
In the past few years emotional neuroscience investigated the neuronal network underlying emotion induction and emotion regulation. Key structures of the “emotional brain” are located within the limbic system, especially the amygdala. Besides these key structures of emotional processing there are additional regions in the prefrontal cortex which are involved in emotional processing [1]. In detail, functional magnetic resonance imaging (fMRI) studies revealed that viewing or evaluating emotional pictures leads to stronger prefrontal activation compared to neutral pictures [2-4]. Using functional near-infrared spectroscopy (fNIRS) an activation of the prefrontal cortex during processing of emotional stimuli has been shown. A first study [5] found increased medial prefrontal cortex activity during an emotional induction paradigm which induces emotions by instructing participants to try to feel like a person whose facial expression is displayed. In accordance with these results, the instruction to remember emotional events leads to an increase of activation in prefrontal brain areas [6]. Furthermore patients with post-traumatic stress disorder show increased prefrontal brain activation to disorder related stimuli [7]. All these studies indicate an involvement of the prefrontal cortex, although its exact function in emotion processing remains unclear. Interestingly, recent studies investigating the neural correlate of emotion regulation processes also described an activation of the prefrontal cortex. Emotion regulation is an important ability to increase, decrease or maintain an emotion on a behavioral, subjective and physiological level [8] which has a great impact on our social life and physical and mental well-being [9]. Recent fMRI studies investigated the specific emotion regulation strategy of cognitive reappraisal – which means that an emotional situation is reinterpreted in order to reduce the intensity of an emotion or to alter the emotion itself [9]. These studies suggest that the process of emotion regulation is mediated by the prefrontal cortex. In detail, the instruction to decrease the effect of negative stimuli by reinterpreting the displayed situation leads to reduced activation in the amygdala and increased prefrontal activation [10-16]. Taking the results of research in emotion induction and emotion regulation together both processes seem to require similar prefrontal regions. It remains unclear whether this prefrontal activation measured with fNIRS is due to emotional induction, emotion regulation or both. Therefore we conducted an experiment in which we can directly compare the prefrontal activation during emotion induction and emotion regulation with fNIRS. fNIRS is an noninvasive optical method which measures cortical activity based on hemodynamic principles. In brief, regional cerebral blood flow (rCBF) increases during neural activation resulting in an augmentation of [O2Hb] and a decline in [HHb] concentration [17]. An increase in [O2Hb] and a decrease in [HHb] can be considered as a sign of neuronal activation. Although fNIRS has some limitations in spatial resolution (and only cortical areas can be measured) we decided to apply this method in this study for several reasons: NIRS is relatively unaffected by movements subjects make during emotion induction or regulation. Additionally, we wanted to validate prefrontal activity during emotion induction measured with this technique.
In sum, the present study consisted of three experimental conditions: emotion induction by viewing fearful pictures, emotion regulation by reappraising fearful pictures and viewing neutral pictures (baseline). With this design we are able to investigate whether the prefrontal brain activation during emotion processing described in previous fNIRS studies were caused by automatic emotional regulation strategies.
METHODS
Participants
Twenty healthy volunteers (mean age 22.4 ± 2.17 years) participated in the study. Only women were tested because of gender-specific differences in the reaction to emotional stimuli [18]. All women had normal or corrected-to-normal vision and were right-handed. Based on self-reports no subject had a history of neurological or psychiatric disorders and nobody was currently under psychotherapeutic treatment or medication. All participants were informed about the nature of the experiment before giving their written informed consent. Subjects gained course credits or 10€ for their participation. The fNIRS investigation of healthy participants was approved by the Ethics Committee of the University of Wuerzburg. All procedures involved were in accordance with the latest version of the Declaration of Helsinki (Seoul, 2008). Before the experiment, subjects filled in the State-Trait Anxiety Inventory [19]; German version: [20].
Stimulus Material
Emotion induction was realized by presenting 80 pictures of the International Affective Picture System (IAPS) [21]. There were two categories of pictures: neutral and fearful. 40 neutral pictures were selected on the basis of normative valence and arousal ratings: these pictures differed least from the mean valence score of 5 (9-point scale, 1 - very unpleasant, 9 -very pleasant) and had the lowest arousal scores (9-point scale, 1 - least arousing, 9 – most arousing)1. Within this neutral category the mean valence score was 5.05 ± 1.72 and the mean arousal score was 2.68 ± 1.94. The selection of the 40 fearful pictures was based on an emotional categorization of the IAPS by Mikels et al. [22]. 40 negative2 pictures were chosen with high ratings (7-point scales, 1 - not at all, 7 - a great amount) regarding the categories “fear” (3.34 ± 1.88), low ratings on the categories of “disgust” (2.34 ± 1.72), “sadness” (2.51 ± 1.65) and “anger” (2.76 ± 1.81) and with high arousal scores based on the 9-point scale of the original normative rating (6 ± 2.16).
Trial Structure
The experiment consisted of three conditions depending on the instruction and the picture category: view-neutral, view-fearful, reappraise-fearful. 40 pictures were presented within each category so that the fearful pictures were displayed twice. This was necessary because there were too few pictures within this category fulfilling the above described criteria. At the beginning of each trial the picture was presented with the instruction word “view” or “reappraise”. The instruction word was placed at the upper edge of the picture, thus not covering any important details of the picture. After 1s the instruction word vanished and the whole picture was displayed for 5 s. After that a black screen appeared for 500 ms and then the picture was presented a second time for 2 s. This second picture presentation was used for a simultaneous ERP measure not reported here. At the end of each trial, participants rated the arousal of the pictures on a 9-point scale (1 - least arousing, 9 - most arousing). A black screen was displayed for 2 s between each trial. We used an event-related design and the order of the trials was pseudorandomized so that the same fearful pictures were not displayed consecutively.
Emotion Regulation Task and Procedure
The training and instruction methods described as follows were previously successfully used by Hajcak et al. [23] and Ochsner et al. [14]. The experiment began with an instruction and training period. The instruction was displayed on a computer screen. If the word “reappraise” was presented at the beginning of each trial, participants were asked to reinterpret the content of the picture so that it evoked less emotion. They were instructed that they could think of a positive outcome of the scene or that the scene was not real. Additionally, the participants were instructed neither to replace their emotion by another emotion nor to think of something different but to dampen their existing emotional feeling. On the other hand, if the word “view” was displayed they were instructed to intensively view the picture and to act out their emotions. After these instructions ten trials consisting of negative IAPS pictures, which were not included in the experiment, were presented. After every reappraisal-trial of this training period the investigator asked the participant to describe her regulation-strategy and, if necessary helped her to generate one. When the investigator was sure, that the subject understood the two instruction types and were able to apply them, the experiment started.
Functional Near-Infrared Spectroscopy
The fundamentals of fNIRS are described in detail elsewhere [e.g. [24, 25]]. Measurements were performed on a continuous wave system (ETG-4000, Hitachi Medical Co., Japan) using a 3 x 11 optode probe set (consisting of 16 photodetectors and 17 light emitters) resulting in a total of 52 channels. The probe set of 52-channel fNIRS was placed over the frontal and frontotemporal area with its lowest-row center optode at the electrode position FPz, extending symmetrically toward positions T3 and T4 (not exactly terminating on these positions because of the fixed interoptode distances) according to the international 10-20 system for EEG (for the craniocerebral correspondence, see [26]). Two different wavelengths (695 ± 20 nm and 830 ± 20 nm) are used by the system, and its frequency is modulated for wavelengths and channels to prevent crosstalk. Reflected light (not absorbed) leaving the tissue is received by the photo-detectors and transmitted into a set of lock-in amplifiers which are limited to the particular frequencies of interest. Both wavelengths are used to solve the modified Beer–Lambert equation for highly scattering media that allows estimating changes in deoxygenated hemoglobin concentration [HHb] and oxygenated hemoglobin concentration [O2Hb] based on the measurements. Since continuous wave systems cannot measure the optical path length [24] and no specific value for the optical path length is adopted from literature [e.g. [27]], the scale unit is molar concentration multiplied by the unknown path length (mmol x mm). Thus, an absolute quantification is not achieved (systems that enable to measure or estimate the path length are described, e.g. in [27] or [28]). The interoptode distance was 30 mm, which results in measuring approximately 15-25 mm beneath the scalp [29, 30]. Sampling rate was set to 10 Hz. Increases in [O2Hb] and corresponding decrease in [HHb] can be interpreted as a sign of functional brain activation [24], but it has to be taken into account that the decrease in paramagnetic [HHb] is the primary source of BOLD-contrast increases [31-33].
fNIRS Data Analysis
The data were analyzed using a general linear model as described elsewhere [34]. We used a combination of a Gaussian function (15 s duration, peak at 7.5 s, dispersion 2 s) and a Boxcar function (5 s duration) to adjust the haemodynamic response function (HRF) to the process of emotion regulation which took place within the picture presentation of 5 s. The peak of the new HRF can be described as a plateau of 5 s. The three conditions “view-neutral”, “view-fearful”, “reappraise-fearful” served as predictors for this HRF. Three linear contrasts were defined on the individual level to test the specific effects of the three conditions: reappraise-fearful vs. view-fearful, reappraise-fearful vs. view neutral, view-fearful vs. view-neutral. These differences were tested against zero using a one sample t-test. Significant cortical activation was indicated by positive t-values for [O2Hb] and by negative t-values for [HHb]. To take account of multiple testing, all statistical inferences were based on an adjusted α-level. This was realized by using Bonferroni correction and the calculation of the Dubey/Armitage-Parmar α-boundary, which included the spatial correlation among the channels ([O2Hb]: r = .27 for the contrast reappraise-fearful vs. view-fearful, r = .17 for the contrast reappraise-fearful vs. view-neutral, r = .21 for the contrast view-fearful vs. view-neutral; [HHb]: r = .22 for the contrast reappraise-fearful vs. view-fearful, r = .16 for the contrast reappraise-fearful vs. view-neutral, r = .22 for the contrast view-fearful vs. view neutral).
Probabilistic Registration of fNIRS Channels to MNI Space
To estimate correspondence between channels and cortical topography, Tsuzuki et al.’s [35] virtual registration method was applied. This method uses structural information from an anatomical database [26, 36] to provide estimates of the channel positions in a standardized stereotaxic 3D brain atlas (Montreal Neurological Institute coordinate system, MNI) [35, 37]. It also estimates the spatial uncertainty due to inter-subject variability of the channel locations.
RESULTS
Arousal Rating
Subjects´ mean arousal ratings were analyzed by repeated measures ANOVA with the factor “condition” (view-neutral, view-fearful, reappraise-fearful). Greenhouse-Geisser correction was applied to the degrees of freedom. This ANOVA revealed a significant effect (F1.38, 26.28 = 56.26, p < .001, ηp2 = .75). Subsequent paired t-tests showed that arousal ratings for “view-fearful” (M = 5.29, SD = 1.56) were significantly higher than for “view-neutral” (M = 2.11, SD = 1.06; t19 = 8.21, p < .001). So, emotion induction took place on a subjective level. Furthermore, fearful pictures were rated as less arousing in the reappraisal condition (M = 3.77, SD = 1.05) than the view condition (t19 = -6.05, p < .001). This reveals that subjects were able to successfully regulate their emotional arousal.
fNIRS Data
The analysis of the contrast view-fearful vs. view-neutral revealed a significant increase in [O2Hb] concentration for channels #8, #19, #37 and #49 (t19 > 3.54, p < .0023) and a significant decrease in [HHb] concentration for channels: #8, #36, #39, #46 and #49 (t19 < - 3.52, p < .0024) indicating activations in left (#8, #19, #39, #49), middle (#37) and right PFC (#36, #46) (Fig. 1). The left hemisphere channels #8 and #19 are probably located (according to the probabilistic registration to MNI space) above precentral lobe and above the triangular part of the inferior frontal cortex, the left channels #39 and 49 as well as the right channels #36 and #46 above the orbitalfrontal cortex. The channel #37 is probably located above superior medial cortex.
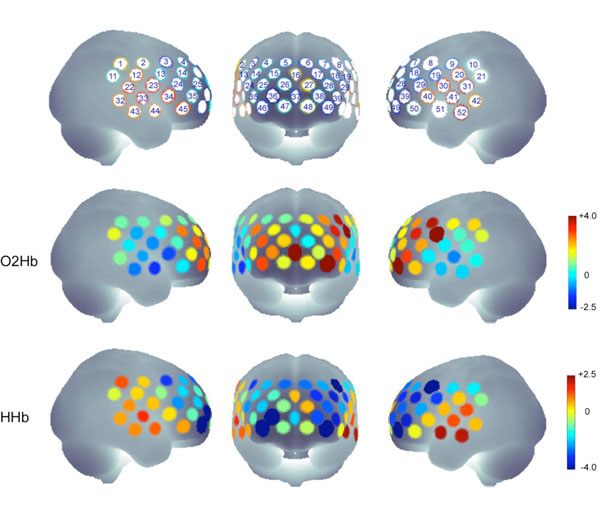
The upper row indicates the channels belonging to the target regions and the probe set definition. The probe set of 52-channel fNIRS was placed over the frontal and frontotemporal area with its lowest-row center optode at the electrode position FPz, extending symmetrically toward positions T3 and T4 (not exactly terminating on these positions because of the fixed interoptode distances) according to the international 10-20 system for EEG.
The middle and lower rows show the t-values for all channels in the contrast “view fearful > view neutral” for O2HB and HHB.
The comparison between reappraise-fearful and view-neutral showed a significant decrease in [HHb] for channels #6, #8, #49 (t19 < -3.64, p < .0019) located in left PFC (superior medial and orbitofrontal cortex). There was no significant increase in [O2Hb] (Fig. 2).

The upper row indicates the channels belonging to the target regions and the probe set definition. The probe set of 52-channel fNIRS was placed over the frontal and frontotemporal area with its lowest-row center optode at the electrode position FPz, extending symmetrically toward positions T3 and T4 (not exactly terminating on these positions because of the fixed interoptode distances) according to the international 10-20 system for EEG.
The middle and lower rows show the t-values for all channels in the contrast “reappraisal > view neutral” for O2HB and HHB.
We could not find any significant activation for the contrast reappraise-fearful vs. view fearful neither in [O2Hb] nor in [HHb] concentration (Fig. 3).

The upper row indicates the channels belonging to the target regions and the probe set definition. The probe set of 52-channel fNIRS was placed over the frontal and frontotemporal area with its lowest-row center optode at the electrode position FPz, extending symmetrically toward positions T3 and T4 (not exactly terminating on these positions because of the fixed interoptode distances) according to the international 10-20 system for EEG.
The middle and lower rows show the t-values for all channels in the contrast “reappraisal > view fearful” for O2HB and HHB.
Correlations
Significant channels within the comparisons view-fearful vs. view-neutral and reappraise-fearful vs. view-neutral were tested for correlations with arousal ratings, state and trait anxiety. Because significant channels resemble difference scores between view-fearful and view-neutral, and between reappraise-fearful and view-neutral, we also computed difference scores for the arousal ratings. We could find one significant Pearson correlation coefficient for the comparison view-fearful vs. view-neutral. A decrease in [HHb] for channel #46 correlated negatively with the state anxiety score before the experiment, indicating that the higher the state anxiety score was before the experiment, the higher the activation was in channel #46 (r = -.58, p = .008). There were no significant correlations between cortical activation and arousal ratings (Table 1).
Correlations Between Significant Channels within the Contrast View-Fearful vs. View-Neutral with Subjective Data for [HHb] and [O2Hb]
| [HHb] | #8 | #36 | #39 | #46 | #49 |
|---|---|---|---|---|---|
| STAI state | -.05 | -.19 | -.38 | -.58** | -.07 |
| STAI trait | -.12 | -.04 | .02 | .00 | -.14 |
| Arousal Rating | .18 | -.21 | .03 | .04 | .17 |
| [O2Hb] | #8 | #19 | #37 | #49 | |
| STAI state | .10 | .03 | -.21 | .19 | |
| STAI trait | -.01 | .29 | .02 | .27 | |
| Arousal Rating | -.15 | -.10 | -.34 | .03 |
** p < .01
DISCUSSION
The main research question of our study was whether prefrontal activation during processing of emotional stimuli - measured with fNIRS - might be caused by automatic emotional regulation processes. First of all we were able to replicate that the processing of emotional stimuli leads to bilateral prefrontal cortex activation. In detail, there was a significant increase in [O2Hb] concentration and a corresponding decrease in [HHb] concentration in bilateral prefrontal cortex (left precentral lobe, the triangular part, the left and right orbitofrontal cortex, and the superior medial cortex) while viewing fearful pictures compared to viewing neutral pictures. The orbitofrontal and the medial frontal cortex has been shown to be involved in emotional stimuli processing and regulation [38, 39].
While the increase of [O2Hb] was seen mainly over left and middle prefrontal brain areas, the decrease of [HHb] was located over the left and right prefrontal cortex. These results support previous fNIRS studies which also reported increased prefrontal activation due to emotion induction [6, 7, 40]. Secondly, we found that this pattern of prefrontal activation could also be observed during an emotion regulation condition, with higher activity during the regulation condition compared to neutral picture viewing condition. This activation was mainly seen over the left prefrontal cortex (superior medial and orbitofrontal cortex), with decreased [HHb] and no changes for [O2Hb]. In contrast to the fMRI literature [10-16] we did not find any significant differences between emotional induction condition and emotion regulation condition. There are two possible interpretations of this missing effect. First, fNIRS might not be suitable to detect the small differences in functional brain activation between the two conditions due to a bad spatial resolution. This interpretation would explain why our results are not in line with several fMRI studies which indeed report prefrontal activation during reappraisal compared to an emotional viewing condition [10, 11, 14-16]. In the same way we could argue that fNIRS measurement is restricted only to the upper 1.5 cm [30] to 2.5 cm [29] of the cortex and is therefore not suitable to measure changes in brain activation in deeper brain structures, for example the anterior cingulate cortex (ACC), which is also relevant for emotional regulation [10, 11, 16]. Alternatively, one may argue that our subjects did not apply emotion regulation strategies at all or in a similar way in both conditions although they were differentially instructed. However, the subjective data contradict this interpretation: subjects rated their emotional arousal significantly lower after reappraisal compared to the emotional viewing conditions, and therefore they regulated their subjective emotional state and did so selectively in the regulation condition. In contrast to the divergent findings of our study and the emotion regulation literature, our data fit very well to a recent study investigating bottom-up and top-down processes in emotion generation [41]. This study found that bottom-up processes, elicited by simply viewing emotional pictures, leads to an activation of the right inferior frontal cortex, the brain area which is in our study selectively activated in the emotional picture viewing condition. This right prefrontal activation might be related to attentional processes, mediated by the parietal cortex, which recruits prefrontal mechanism to direct attention to the stimulus [42]. During the top-down emotion generation condition (viewing a neutral picture and reinterpreting this picture in a way that makes them feel negative) Ochsner and colleagues [41] found an activation of the left prefrontal cortex. Although our reappraisal condition differs from the top-down emotion generation condition in some ways, we also found an activation of the left prefrontal cortex during the reappraisal condition. This left prefrontal activation, especially of the triangular part of the inferior frontal gyrus, might be caused by subjects’ subvocalization to manipulate their negative affect which was present in both conditions (the reappraisal condition and the top-down emotion generation). This is in line with studies showing that the selection and retrieval of semantic knowledge [43, 44] and subvocalization of chronometric counting activate the left prefrontal cortex [45]. As we could not find significant differences between our emotional viewing condition and emotion regulation condition this interpretation of our data is only speculative and must be considered with caution. Taken together, our data support previous studies reporting an activation of prefrontal brain areas during emotional processing [6, 7, 40]. This prefrontal brain activation measured with fNIRS was, at least in our study, probably not caused by automatic emotion regulation strategies, as we did not find significantly different prefrontal activation in a condition in which we directly instructed to reappraise the displayed stimuli, compared to the condition in which we instructed the subjects to simply look at the stimuli. The hypothesized psychological effects of attention for the right prefrontal activity and semantic processing for the left prefrontal activity should be investigated in more detail in further studies.
ACKNOWLEDGEMENTS
Supported by the Deutsche Forschungsgemeinschaft (Research Training Group 1253/1 (Emotions); and the Interdisciplinary Collaborative Research Centre "Fear, anxiety, anxiety disorders" (SFB TRR 58, project B1 and C4))
- Numbers of neutral IAPS pictures: 2190, 2381,2880, 2890, 5500, 5510, 5520, 5530, 5534, 5731, 5740, 6150, 7000, 7002, 7004, 7006, 7009, 7010, 7020, 7025, 7030, 7034, 7035, 7040, 7050, 7080, 7090, 7100, 7140, 7150, 7160, 7170, 7175, 7185, 7187, 7233, 7235, 7490, 7491, 7950.
- Numbers of fearful IAPS pictures: 1300, 1301, 1302, 1321, 1930, 1931, 2110, 2120, 2681, 2682, 2692, 3022, 3500, 3530, 5940, 5970, 5971, 5972, 6190, 6200, 6210, 6211, 6213, 6230, 6244, 6250, 6260, 6300, 6313, 6350, 6370, 6410, 6550, 6610, 6800, 6830, 6940, 9080, 9404, 9630.


